How to Boil Water
❉ In breaking news, scientists have figured out how to boil water – at least 3 times more efficiently and producing twice as much steam. Before you shake your fist at “wasteful research spending”, this isn’t really about your whistling tea kettle!
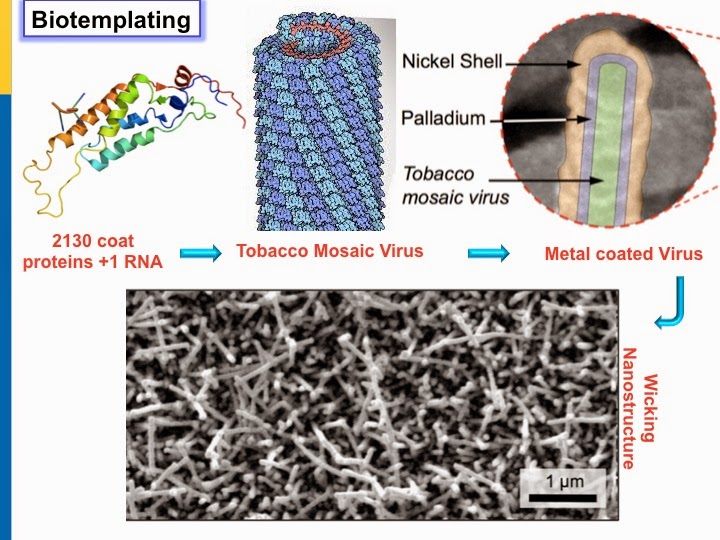
❉ Phase change heat transfer processes (boiling, condensation) are a big part of everyday technology from water purification and HVAC units, power plants and cooling electronics. When water boils, a thin layer of steam can coat the heated surface, insulating it and drastically cutting down on the efficient transfer of heat to liquid. This can lead to surface burnout and a destructive condition known as critical heat flux. What is needed is a surface that discourages the vapor from sticking and wicks in water to quickly re-wet the heated surface. To create a superhydrophilic wicking surface, Drexel University scientist Matthew McCarthy turned to biotemplating with….viruses!
❉ The tobacco mosaic virus causes mottling of tobacco leaves, as its name implies, but is harmless to humans. It was the first virus ever to be discovered (in the late 1880’s) and is constructed simply of repeating units of a coat protein, wrapped around a single, helical strand of genetic material (RNA). A few tobacco plants can produce billions of virus particles, so it’s cheap to make. Dr. McCarthy tweaked the coat protein so it sticks to any engineered surface- from silicon to steel. After dunking the surface in a viral broth, nickel and palladium are added to grow a metallic grass.
❉ The viral tendrils work like a wicking surface, drawing down water to replace what’s boiled away. It’s the same idea behind thermal fabrics designed for athletes which draws moisture away from the body. They say a watched pot never boils. I’d volunteer to test a virally coated tea kettle, how about you?
Waterproofin’ with Hydrophobin: This old post shows how a fungal spore protein can do the opposite, creating a superhydrophobic surface that repels water but allows gases to exchange.
https://plus.google.com/u/0/+RajiniRao/posts/bf9gVFkaTxQ
News Story and Short Video: http://drexel.edu/now/archive/2015/March/TMV-heat-transfer/
Ref: M.M. Rahman, E. Ölçeroğlu, and M. McCarthy, “The Role of Wickability on the Critical Heat Flux of Structured Superhydrophilic Surfaces”, Langmuir 2014, 30 (37), pp 11225–11234.
#ScienceSunday

This is huge.
Bill Carter I was thinking about your work on nanomaterials when I read this story. There are all sorts of self assembling and hierarchical nanostructures in the paper. Some very regular.
This could be a major advance in boiler efficiencies.
Yes, indeed Gary Cameron .
Gary Cameron and also in cooling data centers.
Very interesting advancement in technology. Viruses are being used in more and more creative ways every day.
Brought back memories of boilers, steam turbines and (ugh) steam tables! This was a wonderful read. Thank you Rajini Rao
Chandler Walker it was the use of viruses that got me interested, neat isn’t it?!
Sunil Bajpai I don’t know what steam tables are, but I’m guessing they are not fun to work with 🙂
#ImNoEngineer
This could also be used in water-cooled nuclear reactors, if it were stable for a long time. And car engine internals.
This could be a big deal in the power generation industry. If one can increase the efficiency of a power plant by even a little bit, it could save billions of $ and reduce the carbon footprint of a generation plant.
Very cool.
Nuclear reactors were mentioned in the news story, Nick James, Gary Ray R . This coating increases CHC temperature (indication of failure) by about 3-fold which makes for safer operations.
Rajini Rao Yes, the viruses are a key part of this concept. It reminds me, as a scientist, to keep my mind open and think outside of the box more often.
Rajini Rao Thanks for thinking of us! Actually for the microlattice materials you’ve seen we used a very similar process to coat with nickel (actually it is a commercially available solution). I seem to recall that it is possible to grow amazing amounts of TMV should you decide to – something like 200kg/acre. Talk about scalability! I’m somewhat surprised that the MIT folks (Belcher et al) didn’t go down this road.
Just to note: TMV is also bad for tomatoes and peppers. Possibly the others in the solanacae family. This is why you don’t flick your butts into the garden, and smokers shouldn’t work work on tomato and pepper plants while or after smoking without washing their hands.
Rajini Rao All that Gary Ray R and I need to do now is RTFM:-)
I had been a little bit involved with overheating nuclear plant research and this struck me as a good solution…if it works in that environment.
So are they going to build a new steam engine or furnace for the home?
Now that I actually read the material from Drexel, I am even more impressed.
Here is dry dry dry California, this could make multistage flash desalination type processes more efficient.
Using viruses as nanotechnology factories is brilliant and creative. I wonder what other kinds of coatings and films will come out of this research?
I wonder when they will share this technology #cacti forever.
This could make that big GE gas turbine combined cycle power plant even more efficient. And it already is above 60%.
Yes .but will it bring energy prices down? It seems we’re getting more energy efficient and the costs never wavers.
blanche nonken thank you for that awesome public service announcement re. cigarette stubs in the garden. As you point out, tobacco mosaic virus targets Solanaceae and other families, which include garden favorites such as potatoes, eggplants, pepper, cukes, apples etc. It never occurred to me that a careless cigarette stub could infect them. From the link below, Tobacco mosaic virus is the most persistent plant virus known. It has been known to survive up to 50 years in dried plant parts. Therefore, sanitation is the single most important practice in controlling tobacco mosaic virus.” So, thanks!!
http://theresamccuaigblog.com/2014/05/07/tobacco-mosaic-virus/
Bill Carter what’s also cool is that the Tobacco plant is great for growing transgenic proteins, like the experimental Ebola vaccine that was in the news a few months ago. A great way for farmers to repurpose a cash crop for something healthier.
cc Chandler Walker 🙂
Karen Lewis , no need to design new machinery, just coat the surfaces of existing ones 🙂
I had to share this to our little Materials Science on G+ community, Rajini.
Thanks for finding this.
I think particle acceleration will be the next big thing in energy efficiency.
You’re welcome, Gary. I was thinking of shooting off an email to Dr. McCarthy. Perhaps he will stop by with comments 🙂
Omega742000 Energy efficiency probably helps slow the rises, but the rises often happen for unrelated reasons. Such as: they can.
At least this is good for climate change effects.
Over on Materials, we have posted about the exact opposite type of surface, a superhydrophobic surface (that repels water) almost looks the same, a highly textured surface.
Couple of example references.
Studies reveal new way to make superhydrophobic surfaces with better self-cleaning capabilities
http://phys.org/news/2012-08-reveal-superhydrophobic-surfaces-self-cleaning-capabilities.html
Laser-generated surface structures create extremely water-repellent metals
http://www.rochester.edu/newscenter/superhydrophobic-metals-85592/
So this just improves the heat transfer? It almost sounds like you get something extra for nothing. It still takes x calories to boil x Q water, correct?
Gary Ray R yes indeed, thank you for pointing this out. The contrast with superhydrophobic surfaces (did I spell it right this time, Chad Haney ?!) is very appropriate! I spent some happy hours understanding Wenzel and Cassie-Baxter states and angles on my previous shares on this topic.
One of them is linked in the post above, and the other, I link here only because I am still amused with the wording of Fakir balls (I’m easily amused by word play, as you know):
Lotus Leaves, Rose Petals and Nano-Soccer Balls: What the Fakir could these have in common? (No, I’m not using a bad word, check out the title of the respectable PNAS paper below 🙂
https://plus.google.com/u/0/+RajiniRao/posts/HAsSkbb91RT
P.S. The Univ. of Rochester (my alma mater) research is new to me, thanks for the link!
Note: I noticed the date didn’t seem right for the discovery of TMV so I looked it up. Tobacco mosaic disease was identified in 1886, but it wasn’t recognized as a virus until 1930.
Jim Carver this must decrease the energy used by reducing wastage caused by inefficient heat transfer. Water still boils at the same temperature, but the amount of energy used to convert a given volume of water into steam will depend on the efficiency of heat transfer (the type of conducting material, dissipation into the surroundings, etc.).
Jim Carver Yes, it ‘just’ improves heat transfer – you still get the same amount of heat added to the boiling water. However, this can help efficiency by reducing the amount of heat wasted while doing it – just imagine a kettle on a fire, for instance. If it accepted heat more efficiently, it would boil quicker.
It also, as Gary Ray R and Rajini Rao noted, improves the stability of the heat transfer process and thus increases safety margins. Or, lets you do the same for less with the same safety margins.
Edit. Ha! Like wot Rajini said, but without her eloquence:-)
Can’t wait to see this tech incorporated into a 21st century steam locomotive! Oh, and it’s going to fly, right?
The safety margin effect is a good one, Nick James . My high school physics is so rusty. The heat of vaporization is a constant (which is what Jim Carver is alluding to). To boil one kg of water that is already at 100oC, one needs 1 kg * 2261 KJoules/kilogram = 2261 kJoules.
But that assumes perfect heat transfer, right? If the latter is not perfect, and there is energy loss or superheating, more kJoules will be need to convert that 1 kg into vapor.
Nick James Rajini Rao Right, got it, efficiency of transfer, I thought so, but wanted to make sure. Doesn’t actually lower the boiling point or anything. Thanks 😉 (and I did read the article also)
Great News !
Helped me understand what was going on too, Jim Carver, thanks for the question.
Rajini,
That is another interesting post about “evaporating colloidal fakir drops” (that is a mouthful). If you don’t mind I am going to share that post to Materials tomorrow.
I did not know about Wenzel’s model or the Cassie–Baxter model of wetting surfaces. More to read.
I still can’t spell superhydrophobic or superhydrophilic without looking them up though.
I’ll reread that post and join in the conversation tomorrow, Gary.
I kept misspelling superhydrophobic even after it was corrected by Chad Haney 😀
Rajini Rao Correct, any added energy above 100C at standard pressure is used for the vaporization of the water. That’s why you can’t heat water beyond its boiling point. Or any liquid for that matter. Although you can form a superheated layer at the boundary of the heat source and the liquid interface. This can be a dangerous thing in the lab, so we use things called “boiling stones” in certain cases. I’m sure you know about those.
Oh yes, the boiling stones work by releasing bubbles of air that nucleate the vapor, right? Are they pumice or some specific material?
It’s been so long ago, I don’t remember what boiling stones are made of.
Rajini Rao Yeah, usually pumice, although any porous rock that doesn’t react with stuff in the beaker will work.
Back in school I had a lab partner and she said oh we forgot to put the stone in and before I could say no, not now! whoosh! Nobody got hurt, but it made a really big mess.
I had to go look up boiling stones as I too forgot what they were made of.
Boiling chips are small, insoluble, porous stones made of calcium carbonate or silicon carbide. These stones have pores inside which provide cavities both to trap air and to provide spaces where bubbles of solvent vapor can form. These bubbles ensure even boiling and prevent bumping and boiling over and loss of the solution.
http://orgchem.colorado.edu/Technique/Procedures/Distillation/Boilingchips.html
I remembered the calcium part but couldn’t remember the carbonate part.
By the by, I’ve heard that many people have been burned by “bumping” after heating their soup up in a microwave. They take it out and wind up with the scalding mixture all over them. Best thing is to let it cool a bit and/or poke a spoon in it while it’s still in there. I like to use those wooden skewers or chopsticks for things like that. Actually I use chopsticks for a lot of things in the kitchen instead of a spoon. They’re great for stir frying and they don’t get hot like metal will.
Chad Haney I got lost…calcium carbonate? That’s the major chemical in limestone. The mineral is called calcite.
I just remembered the porous part 🙂
A good PSA on dealing with hot liquids in the kitchen, Jim Carver .
Jim Carver anyone with a new ceramic mug that heats their tea water in the microwave learns this at least once, the hard way. :)
Edit: oops, I should have known someone else would have beaten me to it. 🙂
Are these viruses alive when plated into the container surface? How do they survive? What is their lifespan? Do they breed and multiply while locked into the container surface? I wouldn’t think viruses are durable like plastic or metal.
blanche nonken Oh right…that’s because it’s smooth . Surface irregularities really help in these cases. Microwaves are really bad about hot spots also, even when you have one with a turntable.
You can even map these out using heat sensitive paper. I had to do that one time when we used a microwave for a process.
I wonder if this can be adapted to prevent mineral buildup in water pipes. I live in an area with very hard water, and the deposits clog up everything from toilet valves to sprinklers. If someone can figure out how to adapt a protein to fix that problem, a lot of us would be very, very happy.
Thanks for the share I work with a group that uses TMV for anti viral properties. Interesting application!
X Rellix the viruses are inactivated once they get the metal coatings. They’re just used as a structural scaffold at this point. But they are made as live viruses by the tobacco plant “factory” and easily extracted from the leaves. The question of durability is an excellent one! I was wondering about that too. Unlike a synthetic material like Teflon also used to coat surfaces, the ‘metallic grass’ coating here has protein at its core. Proteins can degrade over time, although again, the nickel/palladium probably preserves the nanostructures.
J Bennett I found this recent report from MIT on a new nanotexturing effect on metal pipes that prevents scale build up. With the new method, once the nanotexture has been created on the surface, oil or another lubricating liquid is applied to that surface. The tiny nanoscale grooves capture this liquid, holding it firmly in place through capillary action, Varanasi says. Unlike a solid nonstick coating, the liquid can flow to fill any gaps, spread on the surface textures, and can be replenished continually if some is washed away. “Even if there’s mechanical damage, the lubricant can return to that surface,” Subramanyam says. “It can maintain its smoothness for an extended period of time.”
These emerging technologies need to find their way into our homes and lives asap!! 🙂
http://newsoffice.mit.edu/2014/new-surface-treatment-stops-scale-buildup-0121
Rajini Rao That’s interesting…mineralization is quite of problem for some folks, especially on the hot water side. The higher the heat, the more the minerals precipitate. And it’s mostly our old friend calcium carbonate. Lowering the pH can help as do water softeners…they replace the calcium ion with sodium through ion exchange, which doesn’t sound like much help, but the sodium salt stays in solution better.
Rajini Rao Fast work! I wasn’t expecting a progress report until next Friday. 🙂
I’ve entered a permanent state of “wow!” when it comes to science and technology. My grandmother came to town in a covered wagon. My mother remembers upgrading from kerosene lamps to electric lights. I remember when the Polaroid Swinger was an amazing imaging device.
Now I can buy a drone that carries a transmitting video camera and checks its position by communicating with satellites. Wow. Just wow…
Thanks for the info on softening water to prevent scaling, Jim Carver !
Yes, just wow, J Bennett !
As to my quick response, I’m procrastinating on a long list of manuscript reviews and grant writing that needs to get done. I’ll get back to them now 😛
Yeah…sounds like fun. ;~)
See you later Rajini Rao
Maybe then we may not need Petroleum any more. We could go back to the steam engine and save the planet.
Desh Maharaj Still need a heat source and this doesn’t change that, it just makes the process more efficient.
And unless you are powering your computer with solar or wind energy, we are still using steam engines…they are more efficient now and called steam turbines. They drive the generators used to make electrical power.
Wups, forgot hydro.
Interestingly, I think the reverse effect of what Jim said about water softeners is used when making common glass (in principle, anyway). Most glass is soda-lime glass, which mainly consists of silica (SiO2), soda (sodium oxide, Na2O, from sodium carbonate), and lime (calcium oxide, CaO, from calcium carbonate), plus some other stuff. Pure quartz silica has a very high glass transition temperature (a couple thousand degrees F) and requires lots of heat to change the crystalline structure into an amorphous state during vitrification. By adding sodium carbonate, they can lower the transition temperature of the mixture to make the heating process more practical to work with, but that in turn results in the unwanted side effect of increasing its water solubility. To counteract that, they add calcium salts.
This post also reminds me of videos I’ve seen about boiling water on the International Space Station. Because there is no buoyancy or convection currents, all the air collects at the heating interface as one gigantic bubble, which interferes with heating efficiency. But I’m thinking they may not have surface failure due to critical heat flux since they can boil water at lower pressures instead of using high temperatures (they do have easy access to a vacuum, after all :P).
Johnathan Chung Yeah, easy access to vacuum and no pump required. 🙂
Sounds like a great lab to me.
Johnathan Chung You may be the person to ask concerning this: We used to be told that a vacuum in “space” was better than one we can create on Earth. I know LEO still has a fairly high number of molecules per unit volume and density decreases the farther you go out.
Do you happen to know what the quality of vacuum is compared to what we can achieve now on the surface? I can imagine that pumps are far better than back then, but any kind of comparison would be appreciated. Thanks.
noted
Nick James duly noted.
Jim Carver I actually don’t know, but the Wikipedia entry has some good info as a starting point for further investigation. That’s a really interesting question.
This section talks about vacuum ‘quality’ relatively to outer space:
https://en.wikipedia.org/wiki/Vacuum#Measurement
And this one discusses the limitations of using pumps to achieve ultra high vacuums: https://en.wikipedia.org/wiki/Vacuum#Pumping_and_ambient_air_pressure
Johnathan Chung Thank you kindly, sir. 🙂
I found this piece very enlightening Rajini. Thank you for sharing.
So we don’t have to worry future fuel or energy
There is a principle in quantum mechanics that says, and I will paraphrase it without the mathematics. So here goes:
The shorter the time a particle spends at one given location, the greater the heat transfer.
I always wondered if that is intuitive. To me it is.
(Particle being accepted in most cases as a molecule, or a set of molecules. Point being in the general case as being a particle.)
Jim Carver Also, Think of how much energy we can have from just trying to box in a particle. The exertion of Energy would be immense
Omega742000 That’s really the principle behind thermoelectricity, but I don’t talk about it much anymore.
Jim Carver The difference can be calculated. But I wouldn’t compare it ,being that space is constantly expanding in fact speeding up;
I am mesmerised by this thread. Before I go to read more about the original article, can someone clarify… The TMV
is a passive structural part of this process, not actively driving it?
(And thanks James Crook for highlighting it to me).
Mags Amond good to have you join the conversation. By active, if you mean is the virus still “alive” and dividing- no. But it certainly drives the formation of the regular nano-sized structures on the surface. Does this make sense?
Jim Carver You should.Particle Acceleration is the Future in Energy efficiency . Not to take away from this article which is astonishing in the way the Virus is basically a better way of energy conservation.
Hey Rajini Rao , this is funny…I still had the Tab open from the article and the image of “Hierarchical structures comprised of virus-templated nanostructures conformally coatings silicon microstructures. ”
And I was just clearing some of the Tabs and thought, hmm, coconut clusters. ;D
It’s been Monday all day over here, hope yours was better. 🙂
Hey Jim Carver my day has been good so far, got a lot done on my To Do list, I think I’ll pack up and take a break now 🙂
Here ya go, Rajini Rao
https://plus.google.com/u/0/+ChadHaney/posts/WC7qwnACd8t
Haha, wish I could plus that again Chad Haney . Installed ToDoist on my Chrome recently so I can collect “karma” points for checking things off my list.
Rajini Rao, I had to divide my ToDo list into ‘Today’, ‘short term’, and ‘long term’. Otherwise, it’s demoralizing how long it is.
I have mine at, ‘Do this right now or there’s hell to pay’, ‘critical, but they wont shoot you, yet’ and meh. 😉
I call it my Too Due list as I never seem to get it all done.
These are all hilarious and practical suggestions. Now I don’t feel bad about writing down a task that I’ve already done so I get to cross it off.
Rajini Rao thanks for sharing this interesting news.
Hiiii Rajini Rao
Good
Rajini Rao Yes – thank you.
good nice
+Rajini Rao What are the consequences? A decrease in the boiling point temperature or of the latent heat of vapourisation? Or something else?
massimo valerio Preite the consequences include a reduction of energy consumption due to increased efficiency of heat transfer at the heating surface, and also an improved safety margin. See explanations in the comments above.
Does this happen naturally anywhere on the earth?
D L Clark great question! Both superhydrophilic and superhydrophobic surfaces are found in nature, for example, on plant surfaces. Mosses absorb water to many times their weight using similar surfaces. Other examples are epiphytes, that grow on trees (orchids) and even carnivorous plants that use such surfaces to trap insects. Check out sections 3-4 of this paper: http://rsta.royalsocietypublishing.org/content/367/1893/1487
Dalo by se použít tuto schopnost pro PC procesory, pro lepší odběr tepla: -)
Distilled water
What is absolute temperature
Really interesting