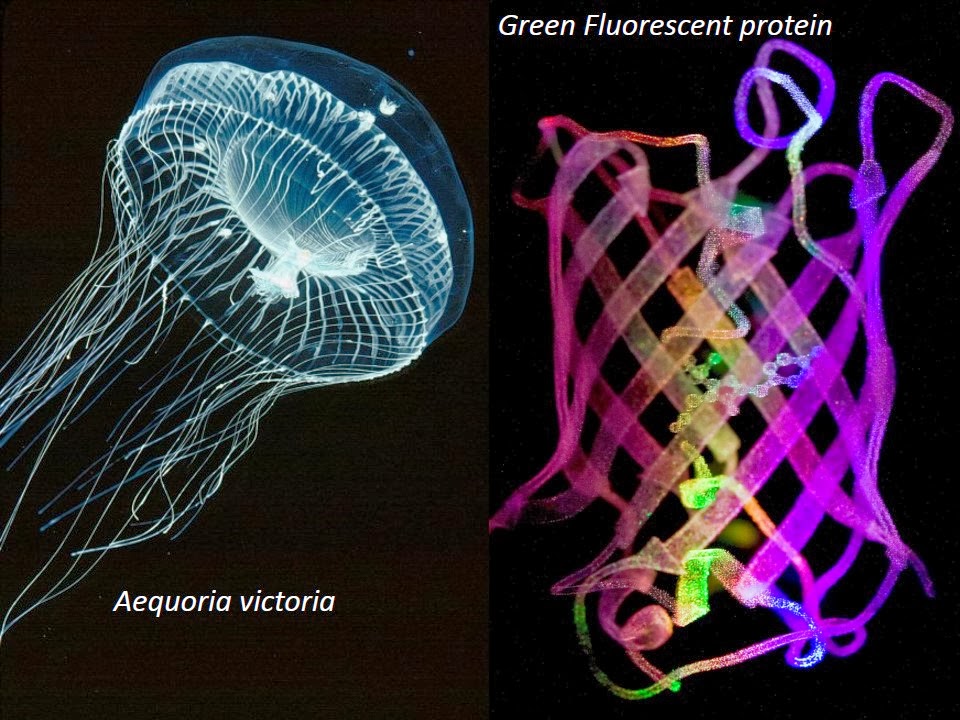
Jellies to GFP
The Story: Drifting along the currents off the west coast of N. America, the jellyfish Aequoria victoria gives off bursts of blue luminescence when a protein called aequorin binds calcium. The blue light is absorbed by another protein with the unimaginative name of green fluorescent protein (GFP), and the jellyfish now glows…surprise…green! After scientist Osamu Shimomura first observed that GFP could fluoresce independently of any added factors, Doug Prasher cloned the GFP gene and worked out its DNA sequence. At Prasher’s seminar, Martin Chalfie realized the potential of GFP: he could insert the gene behind any promoter (on switch) and the cell would glow green when the gene was turned on. Prasher handed out the cloned gene to Chalfie and hundreds of labs. This led to a multitude of uses of GFP, now a favorite workhorse in any research lab. Roger Tsien tinkered with the gene to generate a rainbow of different colors. Sadly, when Shimomura, Chalfie and Tsien were awarded a Nobel (in 2008) for their work, Prasher was conspicuously omitted, because no more than 3 can share the prize. Worse, he was out of a job by then, driving a courtesy shuttle bus for Toyota (apparently he is back in lab now). Still, Prasher expressed his delight over the Nobel, and he was invited to the ceremony and publicly thanked by his more fortunate colleagues.To paraphrase Newton, if we have seen further, it is by standing on the shoulders of giants.
The Science: GFP is built like a barrel of crisscrossing ribbons known as beta strands. Nestled in the middle, is a strange cyclical arrangement that spontaneously forms from three consecutive amino acids (Serine, Tyrosine, Glycine or SYG in their single letter code). It is this cyclical arrangement that gives GFP the ability to fluoresce. The same sequence in other proteins does not cyclize or fluoresce. Adjusting the environment around this changes the spectral properties (color), stability (so it can be used in warm blooded animals), and regulation (by pH or ions).
The Gift: Oh GFP, how do I love thee? Let me count the ways. Care to snoop on the business of your favorite protein? Tag it with GFP and become a video voyeur, watching it move in a living cell. Want to measure the pH (acidity) of a cell? There’s a pH-sensitive GFP mutant named pHluorin for that. Curious if two proteins interact? Tag one with YFP (glows yellow), another with CFP (glows cyan). When they come together, the emission from CFP activates YFP and you get a yellow signal. Create a Brainbow by genetically mixing GFP variants so each neuron is colored differently. Can you blame me for the bad poetry? 🙂
Pop Sci: http://imgur.com/gallery/krNEH
Nobel Lectures: Follow the links in http://goo.gl/51d40L #ScienceEveryday

Why hasn’t this gene been added to human skin cells, cultured and used as body art yet?
Whoa, that might actually work, Richard Healy . Sort of like a fluorescent tattoo 🙂
Rajini Rao It may bleed and become splotchy over time. That’s something that probably will need experimentation alongside mixing with tattoos and whatnot.
[edit: the color may bleed]
Also, unfortunately, the fluorescence will be transient. The DNA introduced into skin cells will only last for a couple of weeks with current technology. The only way we can make it permanent is to insert it into the germline (designer babies!). They do this with fish and bunnies, but I’ve not heard of human volunteers 😉
Rajini Rao That could be a good thing. It would avoid the whole tattoo remorse thing and make for repeat business as people return to the parlor to renew their art.
This idea is getting better 🙂
Jellyfish are so mysterious Rajini Rao !! They have no brains but scientists are baffled as to how a certain group of jellyfish remember to travel to the same feeding location.
Bobby Ryan , I’d not thought of their migration. I wonder if they are sensing currents or magnetic fields and how?!
Awesome post Rajini Rao ….you make science look so cool and simple for people who think science is boring and complex…..
Is so beautiful, nature is amazing!!!!!
Thanks, Kashif Ansari . Science is sometimes complex but hopefully never boring 🙂
And then there’s phototoxicity. Great share. A critical tool for modern biology.
Hi Scott Sneddon , do you mean phototoxicity to selectively kill cells that express a fluorescent protein? There is Killer Red, a fluorescent protein that appears to be a GFP variant, that can be activated by light to release toxic free radicals and surgically kill the cell that expresses it. It has potential in photodynamic treatment of cancer, for example.
I found this paper (open access): http://www.jbc.org/content/284/46/32028.full
Yes, I was referring to the general phototoxicity of inducing fluorescence in living cells. Makes me a bit wary of the GFP tattoo 😉
Turns out that most GFPs are less phototoxic than other fluorescent molecules because the chromophore is buried, so fewer reactive oxygen species seem to be formed on prolonged illumination. Killer Red is an exception, and of course it’s been put to good use as you point out.
Another source that discusses GFPs generally, and exactly how the protein arranges the amino acids to create the fluorescence can be found at
http://zeiss-campus.magnet.fsu.edu/articles/probes/fpintroduction.html
Thanks for the link, Scott Sneddon . I’m saving it for reference..very comprehensive guide to GFP. Another (minor) caveat to its use is that GFP takes some time to fold and develop fluorescence because of the complex cyclization of the chromophore. So it’s not a good idea to use it to follow biosynthesis of your favorite protein since the appearance of fluorescence may be delayed relative to the synthesis of the tagged protein. Probably not good for tracking protein turnover either, since the GFP barrel hangs around in lysosomes for quite a while, after it has been clipped off the target protein. Of course, it is great for tracking the localization of the target, once it has been synthesized.
Fantastic post, Rajini, informative and entertaining!
More useful information Rajini Rao. If you’re interested in trafficking, that’s a pretty crucial caveat (though the pHluorins can help untangle at least part of the pathway) .
Sharing this post to my Bookmarks Circle for later reference.
While we’re at it. Even the faster maturing “turbo GFPs” take ~20 minutes to develop fluorescence versus about an hour for EGFP, so the caveat still applies.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1618374/
and
http://www.evrogen.com/protein-descriptions/TurboGFP-description.pdf
Scott, I had not heard of the Turbo version, thanks! 🙂
Thanks for the interesting read Rajini Rao
Just sad Prasher was omitted from the prize.
Same here, Susana M. . He sounds like an all round nice guy. Cloning and sequencing genes back in those days was a big deal, yet he freely gave away his clone.
Great post. I was just telling a group touring my facility about how GFP and luciferase are powerful tools in cell signalling research.
Sir I have eating moon jelly fish for years thy don,t taste good but seam to fill the void I have herd that tear are some benefits to human brain can you confirm this
Sir? Do you mean Professor Rao, madam scientist?
It may be the effect of consuming all that moon jelly Chad Haney .