Llama Pharma
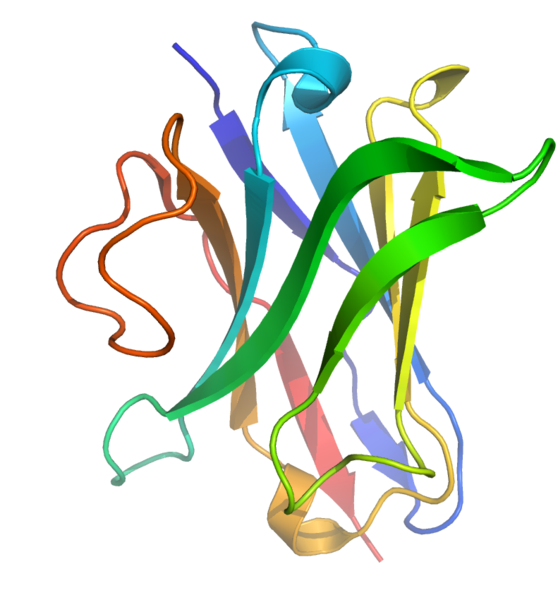
• Camelid Nanobodies for Therapy: Circulating in the healthy immune system of llamas, camels and alpacas is an unusually small version of antibody– proteins that are key to fighting infection. In contrast to our antibodies that are large and cumbersome (120-150 kilo Daltons in size), single domain antibodies made from the Camelid family are only 12-15 kDa or 4 x 2.5 nanometers in diameter.
• Potent and penetrant, these little proteins (right image) are more soluble and stable than their larger counterparts. They can get deep into tissues or cross the blood brain barrier where they have the potential to neutralize viruses, deliver toxins to cancer cells or even fight fungi in formulations of anti-dandruff shampoo. Concerned about alpaca abuse? No worries, they can be produced in bacterial factories.
Sources: (i) http://bpod.mrc.ac.uk/archive/2012/10/4
(ii) http://en.wikipedia.org/wiki/Nanobodies
(iii) Saerens et al., Single-domain antibodies as building blocks for novel therapeutics. http://dx.doi.org/10.1016/j.coph.2008.07.006
#scienceeveryday


Hey, he has got that 1980’s punk hair style … 🙂
Yeah, I like his ‘do! 🙂
Kinda looks like Billy Idol
fungi in formulations of anti-dandruff shampoo, who knew? I wasn’t even aware that M. furfur was implicated in dandruff.
I bet he would love to dance along to the tune of DuranDuran or Bananarama … hihi.
Dandruff is a fungal infection. Zinc pyrithione in most antidandruff shampoos is a potent antifungal..as is tea tree oil. Too much information? 🙂
Russell Bateman oh ya … … Billy Idol … now that’s what this llama looks like.
I knew about tea tree oil, just didn’t pay attention to the anti-fungal part.
Next time, I wash my hair with copper sulfate.
I used too have dandruff, and i don’t use dandruff (zinc or copper) shampoo anymore. I don’t have anymore dandruff nowadays.
Heh, weirdly enough I’d just been reading about camelid immune systems and their weird antibodies. Cool to see that people are messing about with them.
More seriously, what is the downside of it ?
If it was so positive, evolution will have “invented” it already knowing it is a simpler, smaller molecule.
Maybe the proper questions should be, why did it appear in camelid evolution, when did it appear (if we can trace this)
Olivier Malinur , I don’t think that the blue in anti-dandruff shampoo is due to copper sulfate..I’m not seeing copper listed as antifungal. Let me know if you have a link.
Rashid Moore , a bit lost on the connection. Is it the bad hairdo?
John Bump , yes! there is good progress in the field although intestinal absorption of nanobodies is one limiting factor at present.
There are lots of places where evolution messes up: humans can’t make Vitamin C, while all other animals can, for instance — yet we seem to be doing pretty well. Camelids have a bunch of adaptations for operating under extremely dehydrated circumstances. Maybe tiny antibodies work better in very viscous blood. Camelids have crescent-shaped red blood cells, rather than our round ones, to handle dehydration, so similarly changed antibodies wouldn’t be surprising.
Olivier Malinur , check out the Wiki link..you can clearly see the evolutionary connections between human immunoglobulins, camelid and cartilaginous fish (sharks also have simpler antibodies) in the second figure. These are all variations around a common theme. We see this often in evolution. There is nothing surprising about the variants, except that it requires curiosity driven research to discover it.
OK, thanks Rashid Moore 🙂
Mr. Llama may win the cuteness contest, though.
I did not even think about the blue of the shampoo. Copper sulfate is used for vine to fight mildew. Bouillie bordelaise is a classical mix, lime + copper sulfate + water.
Olivier Malinur , okay. I wonder if it is shampoos though.
That makes a lot of sense.
John Bump: “weirdly enough I’d just been reading about camelid immune systems and their weird antibodies.”
. . .
– It isn’t that weird, they just have an extra set of immunoglobulins as a result of a duplication of a gene that encoded for a heavy chain of IgG’s.
Rajini Rao: “In contrast to our antibodies that are large and cumbersome (120-150 kilo Daltons in size), single domain antibodies made from the Camelid family are only 12-15 kDa or 4 x 2.5 nanometers in diameter.”
. . .
– This comparison isn’t quite fair. Camelid-derived nanobodies are synthetic (the original camelid IgG’s from which they have been developed are dimers formed by two heavy chains, they just lack the two light chains present in the other IgG’s) whereas out antibodies are produced by our immune system. There are smaller synthetic versions of our anti-bodies as well. While nanobodies have only one antigen recognition domain, other proteins derived from murine or human antibodies need at least two.
BTW, nanobodies are patented.
Copper sulfate is often used in ponds to control algae. It’s a tricky one though. An overdose of the substance in Twin Lakes, CO caused a fish die-off in the Arkansas River downstream from the lake for many tens of miles in the 1980s. It took many years to recover and altered the natural genetic make-up of the stream since all new fish were stockers.
Oh yeah, I didn’t finish…I don’t think it’s in shampoo because it’s considered a toxic substance for aquatic wildlife.
Can the proteins be present in plasma? If yes, can they be retrieved and placed into a mouse to test if it would fight infection.
My apologies if the above is wrong. I’m not into the same field of study as yours.
Generally copper is used to control any fungal invasion. In aquarium, it is recommended, if fishes have white spots diseases.
However, salt water fishes are very sensitive to copper, so be careful.
It is used also to prevent mildew for tomatoes. My grand dad, who was horticulturist, used to pierce the bottom of the stem of tomato plants with a copper thread, if we knew that mildew was coming this year (usually, rainy hot summer).
Jim Carver , I agree. Copper sulfate is quite toxic. Perhaps the blue in Selson Blue is just a dye.
Shah Auckburaully , you’re right..the proteins are in plasma and can be harvested from it. But, it is more convenient to make them by recombinant DNA methods in bacteria.
Olivier Malinur does potassium permanganate used in aquarium affect copper levels? Coz they are used against infection
Rajini Rao how do you recombine DNA in lab? Is it a complex process?
Olivier Malinur You can, I personally exclude all copper (including plumbing) in my tanks. If you maintain good water quality there is no problem with diseases.
Rajini Rao Pretty sure it is a dye, FD&C Blue no. x or some such…
Shah Auckburaully: “Can the proteins be present in plasma?”
. . .
– Nanobodies are expected to be less immunogenic (so they could be used in larger concentrations and more extended periods of time) and cause fewer problems (e.g., formation of antigen-antibody complexes) than regular antibodies due to their smaller size (fewer epitopes) and the presence of only one recognition domain, respectively.
Currently, many of the antibodies used in immunotherapy have been humanized and some are even totally “human” (although produced in other organisms) to minimize their antigenicity.
Zephyr López Cervilla , notice my careful use of the phrase, “single domain antibodies made from the Camelid family”. I never said that the natural Camelid antibodies were single domain. “Made from” is synonymous with “derived from”, in this context, as is obvious in my reference to manufacture in bacteria further down.
I am well aware that Nanobody is the trade name from Ablynx, since that is the first sentence in the Wiki link that I quoted. Did you think I did not know or are you bringing it up as an interesting factoid for our readers? The latter is always appreciated! However, if you are looking to correct me on facts, I’m all game to play with you, but you do need to go beyond superficialities that are already in the links that I posted 😀
For example, a worthy challenge would be to question how the single domain antibody could be expected to neutralize a viruse or toxin if it is monovalent (natural antibodies are bivalent)?
Shah Auckburaully , I have no idea. Potassium permanganate (PP) is used in “nursery aquarium” not in the main one. At least this is the proper practice.
I have had an aquarium since the age of 16.
When I just started, I used PP to treat some Barbus which were sick (a small arthropod in the gills). I was starting chemistry. I did not have a solution of PP but just powder. But to impress my brother, I made long calculations with avogadro number, molecular mass and idk what bs… At the end, I got a “proper concentration”. I added 5 cl of it in the nursery aquarium which turned deep purple (normal, it was the late 70s…).
Put the Barbus inside. It swam faster than all fish you could see, jumped out of the aquarium. I put it back inside. It NEEDED to stay 3 min. After 3 min, it was floating at the surface.
My brother asked me if I was sure of the remedies. I show him my page of calculations. He trusted me, foolish boy…
We put back the Barbus in the aquarium, it revives and left a purple cloud at the back when swimming. Then, a couple of convulsion… Dead…
I gave my calculation to my chemistry teacher.
I made a mistake of a factor 6022… Thank you mister Avogadro…
Shah Auckburaully , recombinant DNA technology is straightforward and routine! DNA can be specifically cut, as with a scissor, with restriction enzymes that recognize naturally occurring palindromic sequences in the DNA. These fragments can be combined and ligated by other enzymes. Or, DNA can be amplified using a reaction called polymerase chain reaction. Both these techniques were game changers and were recognized with Nobel prizes.
Olivier Malinur That’s a good story!
Not for the barbus.
Of course, Drew Sowersby . Some regions are notoriously hard to amplify or clone because of secondary structures or toxic (to bacteria) sequences. Some techniques may be challenging (single cell or very long PCR). Given that there are commercial kits for all the common steps, and that undergraduates have cloning exercises in class room labs, I would say that in general, recombinant DNA technology is routine.
The recombinant DNA technology mindboggles me.
Is it possible it happened “naturally” and part of some fish DNA passed in a mammal through a bacteria ?
I mean, already, we have some virus genetic material in our DNA…
(Ok… I have the feeling I will be tagged as the “dumb poet”)
Olivier Malinur , I would not say that recombinant DNA technology happened naturally, per se. Restriction enzymes are made by bacteria to chop up foreign DNA..as protection. And you’re right, there are natural viruses that can jump around in our genomes, carrying snippets of information. The technology as we practice it today, comes with our harnessing some natural tools along with some man made manipulation. I’m not sure I answered your question adequately, though 🙂
Actually, Olivier Malinur you’re not too far fetched. This wiki article is good:
http://en.wikipedia.org/wiki/Horizontal_gene_transfer
I agree, Drew Sowersby . I was making site directed mutations in yeast cytochrome c well before there were any kits or even written protocols. The original paper by Zoller and Smith was my only guide. I had to use 32P isotope labeled oligos to fish out my clones by plaque hybridization.
One problem with kits is that people who rely on them have no idea about the steps or components beyond adding Buffer A in Step 1. So when something goes wrong, they have no idea how to trouble shoot.
Good link, thanks Jim Carver . I think that is what Olivier was asking about.
Drew Sowersby , I think Zephyr was responding to a question by Shah and mistyped your name instead 🙂
It happens !!!
This is frightful. So “The Fly” was not 100% wrong.
Olivier Malinur Interesting huh? We are now re-thinking our assumptions about the process of evolution due to this.
Rajini Rao and I talked about this not too long ago.
Rajini Rao: “+Zephyr López Cervilla , notice my careful use of the phrase, “single domain antibodies made from the Camelid family”. I never said that the natural Camelid antibodies were single domain. “Made from” is synonymous with “derived from”, in this context, as is obvious in my reference to manufacture in bacteria further down.”
. . .
– I didn’t say that you were wrong, just that the comparison was unfair, like comparing a mass produced car with a racing car. A fairer comparison would have been nanobodies versus microantibodies, or at least versus an IgG Fab fraction.
__________________
Rajini Rao: “I am well aware that Nanobody is the trade name from Ablynx, since that is the first sentence in the Wiki link that I quoted. Did you think I did not know or are you bringing it up as an interesting factoid for our readers? The latter is always appreciated! However, if you are looking to correct me on facts, I’m all game to play with you, but you do need to go beyond superficialities that are already in the links that I posted”
. . .
– I didn’t try to specifically inform you, I was trying to create awareness of how obvious many patents that are granted in biotechnology (and other fields) really are.
The use of a slightly modified protein that have been found in an organism with the same original purpose (the recognition of specific epitopes) in a different organism is an obvious idea. This patent should have never been granted. Its development was simply a matter of investing enough resources. Not very different from Celera Genomics’ approach at the time the human genome was being sequenced.
As for the factoid, I hadn’t opened any of the links so I couldn’t know their content.
__________________
Rajini Rao: “For example, a worthy challenge would be to question how the single domain antibody could be expected to neutralize a viruse or toxin if it is monovalent (natural antibodies are bivalent)?”
– Simple, making that single domain interact with with the recognition domain of the virus protein that the virus uses to recognize its receptor to infect a cell. Or course, this won’t always work, the virion usually have many copies of this type of protein on its surface, so to effectively neutralize it you need to block most of them and with greater affinity than with its receptor.
With toxins it’s easier since they usually only contain a single interaction domain with its surface receptor.
__________________
Ok, Jim Carver , you made my job half. Now, can this simpler antibody from camelids comes from horizontal gene transfer ?
Olivier Malinur I think that’s possible and actually quite likely although others may disagree. So the question remains, why don’t we have it? Probably because the selection pressure wasn’t there for us, like it was for them.
Drew Sowersby: “Can you clarify the question
+Zephyr López Cervilla? Isn’t plasma full of dissolved proteins?”
. . .
– I wouldn’t use the “full” word (as with any solution there’s a saturation threshold and the physiological concentration is below that level).
There are different proteins solved in blood plasma (globulins), although by far the most abundant is albumin (≈ 65 kDa).
Excellent points all, thanks for the clarification Zephyr López Cervilla . The single domain antibody is smaller than the commonly used Fab fragment IgG (50 kDa) and even smaller than the single chain variant (25 kDa). I think it is fair to say that the camelid version is smaller than its better known counterparts.
I do put some thought into the links in my post. The first is a little blurb of info for the casual reader, the second is easily accessed Wiki page, and the third is a solid peer reviewed article with nice figures.
Finally, one can introduce multivalency by inserting a short linker between identical VHH domains..this way, it can bridge neighboring epitopes and crosslink them together, much the same way that full size antibody does, while still being very small.
Olivier Malinur and Jim Carver , in answer to your musings, here is a paper that addresses “the convergent features of single-chain antibodies that permitted their emergence and reemergence and the constraints that may have precluded their existence in all vertebrates.” I don’t have time to read it right now, but I’m leaving the ref here for anyone who may want to take a look:
http://www.plosbiology.org/article/info%3Adoi%2F10.1371%2Fjournal.pbio.1001120
Drew Sowersby: “Are you trying to give me a semantic neck trim +Zephyr López Cervilla? I see your question was not aimed toward me so nevermind.”
. . .
– That’s right, in my first post I was trying to reply to a previous comment written by Shah Auckburaully but I accidentally misquoted it.
Doesn’t your stream move up and down, making it hard to choose the right option to cite someone? I finally opted to open this thread in an independent tab.
It is difficult for a geologist to understand but I picked the message. Convergence because of urea in shark blood (which is the reason why we don’t stun shark with a club in Caribbean but we bleed them: if you break the back of shark, It tastes like pee ) and very dry environment in camelid’s case. The molecular structure with disulfid bridges helps stability.
Why also monotremes and marsupials? Maybe because they have common ancestors the sphenacodonte which evolved in Triassic, very dry period. Worth to dig…
To my humble opinion, there are good chances that placodermes were similar as well, following the same reasoning.
Olivier Malinur: “Convergence because of urea in shark blood” […] “and very dry environment in camelid’s case. The molecular structure with disulfid bridges helps stability.
Why also monotremes and marsupials? Maybe because they have common ancestors the sphenacodonte which evolved in Triassic, very dry period.”
– I didn’t get the same conclusion after reading that paper. As I see it, the reference of high concentration of urea and extreme temperatures is mentioned exclusively as a possible explanation of the generation of intradomain disulfide bonds in the heavy-chain-chain-only antibodies (HCAb’s) of both camelids and sharks:
<>
_________________
The generation of this kind of HCAb’s would be beneficial for any species. But it is a very rare event due to the organization of the Ig genes on most vertebrates:
<<we believe that they are clearly useful and, anthropomorphically speaking, all species would love to have them; however, the organization of Ig and TCR loci in all vertebrates besides cartilaginous fish precludes, or makes quite difficult, the capacity to generate them. Consistent with the idea that single-domain Vs are a useful innovation, once they emerged in the ancestral camelid and cartilaginous fish species, HCAbs have been perpetuated as a major immune system attribute in all of the descendant species.>>
_________________
Camelids must have been lucky to have generated functional HCAb’s. Camelids didn’t probably “need” HCAb’s more (i.e., they weren’t exposed to stronger selective pressure) than other mammals (or birds, whose body temperature is generally higher and can live in quite arid conditions and also exposed to extreme temperatures). Other vertebrates have been able to survive without HCAb’s in similar environmental conditions.
So probably there’s no link between any of this adaptative convergences other than representing a clear evolutionary advantage for all those organisms.
Maybe … As I said , I am geologist .
Not molecular biologist.
However, the fact that both monotremes marsupials have this HCABs in common is very interesting. Marsupials and eutherians are supposed to have diverge from some monotremes or even multituberculates mid jurassic.
The fact that 2 out of 3 of the Therians have this HCAbs cannot be a coincidence.
I’ve just remembered something about camelids that may be or may not be related with this topic. Unlike the rest of the mammals, the camelid erythrocytes aren’t disk-shaped but rather they have a similar shape to a rugby ball.
If I remember correctly, it is supposed to be an adaptation to life at high altitude (and dehydration).
I must point out that the natural habitat of most of the camelid species (Bactrian camel, llama, alpaca, vicuña, guanaco en.wikipedia.org/wiki/Camel_family) are lands at high altitude (the Andes, the Gobi and Taklamakan Deserts).
The camel dromedary may have arisen rather recently, once the Arabian desert was formed (probably around the same time as the Sahara Desert).
It seems that this shape allows them to have higher hematocrit (the volume percentage of red blood cells in blood en.wikipedia.org/wiki/Hematocrit) without higher risk of suffering a thrombosis caused by an excessive viscosity of their blood.
My guess is that perhaps thanks to be smaller, those H-chain-only antibodies may also contribute to make their blood less sticky, particularly during the period of time of an infection in which the IgG production is highest.
Fascinating info, thanks for the summary Zephyr López Cervilla and Kudos to a geologist (Olivier Malinur ) digesting that paper. The other point I recall about the convergent evolution in the paper is that the camelid translocation (gene rearrangements) happened recently whereas the one in sharks is ancient.
Rajini Rao That tends to imply that the translocation of that gene was ubiquitous over time.
or could have been
The translocation was an accidental event. Some are quite deleterious, and occassionally, beneficial. It would have happened in the camelid ancestor and passed down the llamas, alpacas and whoever. Did I understand you correctly?
I will be interested to see what of the case of monotremes and marsupials.
Rajini Rao No, I think we were talking about two accidental events.
What I meant was that if the interval of time was large enough and the same traits showed up, then it was ubiquitous and was just waiting around for a proper pathway. I’m sure many failed along the way.
We’re just tradin’ cards ya know, the deck is amazingly huge…but it’s finite and they have transferred back and forth in nature.
Olivier Malinur , I was able to track down one free read on the origins of the TCR mu genes (these are the ones like the Camelid antibodies; TCR stands for T cell receptor) in duckbilled platypus (non placental mammal like monotremes and marsupials). The last sentences summarizes their findings: A compelling model for the evolution of the Ig and TCR loci has been one of internal duplication, divergence and deletion; the so-called birth-and-death model of evolution of immune genes.
See, http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3457774/
Olivier Malinur: “the fact that both monotremes marsupials have this HCABs in common is very interesting. Marsupials and eutherians are supposed to have diverge from some monotremes or even multituberculates mid jurassic.”
“The fact that 2 out of 3 of the Therians have this HCAbs cannot be a coincidence.”
– This isn’t quite accurate, monotremes and marsupials don’t have HCAb’s but single-domain TCRs:
<<Single-domain Vs have evolved also in monotremes and marsupials via generation of an entirely new locus, which is a chimera between Ig and TCR loci (Figure 2) [24].>>
—————–Figure 2. <>
(4) TCR-μ
MONOTREME (platypus)
MARSUPIAL (koala)
_________________
Does it really matter? In the case of IgG’s their predominant form is as soluble protein in plasma and other extracellular media (although they also exist as membrane proteins named BCR in B-cells with also an essential function), whereas the TCR is always a membrane-bound protein in T-cells.
So possibly the camelid HCAb’s and the marsupial/monotreme TCR-μ don’t provide the same advantages, unless their common advantage were exclusively related to their function as membrane-bound protein.
In fact, In my view the existence of the TCR-μ receptor even more intriguing since its potential advantages don’t seem as obvious as in the case of the HCAb.
So at the end, can l use blended koala extract to heal dandruff ?
LOLOL, no. But the polyterpenoids in the eucalyptus that the koala likes to eat are pretty good antifungals.
Reminds me of my daughter’s high school science project …she showed that oregano oil was more potent than tea tree and other essential oils in killing fungi. Carvacrol, thymol, eugenol ..all abundant in herbal extracts and great antifungals.
Olivier Malinur Suzanne Catty has some stuff that’s just great for getting rid of scaly skin. It’s called ‘Skin Soother’ it works great all over.
http://www.newworldorganics.com
It’s good stuff, I use it and no, I am not a paid spokesperson. 🙂
Olivier Malinur: “So at the end, can l use blended koala extract to heal dandruff ?”
. . .
– I have to disagree. You can, but you will need to previously perform a similar procedure to the production of nanobodies, that is, the removal a chunk of the protein (in fact it’s the gene what is shortened). In the case of the TCR-μ, its transmembrane domain (and its cytosolic domain). Not a big deal, in fact it’s easier to express that way in say, a bacterial system.
I’ve found this paper in my folders. It contains nice electron micrographs (page 100) and TEM electron micrographs (pages 102 and 104) of camelid erythrocytes:
– Cohen WD and Terwilliger NB. Marginal bands in camel erythrocytes. J Cell Sci (1979) vol. 36 pp. 97-107
ncbi.nlm.nih.gov/pubmed/457820
jcs.biologists.org/content/36/1/97.long
jcs.biologists.org/content/36/1/97.full.pdf
I was right, most of them look like rugby balls, although flattened:
SUMMARY
The elliptical, enucleate erythrocytes of camels have been examined for the presence of marginal bands and their constituent microtubules.
[ . . . ]
The marginal band (MB) is a discrete circumferential bundle of microtubules with a probable role in alteration and perhaps maintenance of cell shape. MBs occur in the elliptical, nucleated erythrocytes of non-mammalian vertebrates (Dehler, 1895; Meves, 1911; Fawcett, 1959), in the thrombocytes of both mammalian and non-mammalian vertebrates (Fawcett & Witebsky, 1964; Behnke, 1965; Sandborn, LeBuis & Bois, 1966), and in blood cells of certain invertebrates (Cohen, Nemhauser & Jaeger, 1977). MBs have not been observed in the mature anucleate, diskoidal† erythrocytes of mammals.
Among the mammals, members of the family Camelidae (camels, vicunas, guanacos, llamas, alpacas) are unique in that their erythrocytes, though anucleate, are elliptical (Andrew, 1965). The question thus arises as to whether MBs play a role in cell shape generation and/or maintenance in these species.
[ . . . ]
† Terminology in the literature is often confusing with respect to erythrocyte shape, referring to ’round disks’, ‘elliptical disks’, and ‘disk-shaped’ elliptical cells. The terms ‘disk’ and ‘diskoidal’ should be reserved for cells which are flattened and circular; a disk cannot be elliptical.
[ . . . ]
Fig. 2. Camel erythrocytes in mammalian Ringer’s solution (dilution of whole blood required for observation of individual cells). Typical cells are flattened, anucleate, and elliptical, with the long axis of the ellipse in the range of 7-9 micrometres. Phase-contrast, x 1420.
[ . . . ]
The occurrence of MBs in camel erythrocytes is possibly correlated with ontogeny of distinctive physiological properties. Camels are adapted to survive extreme dehydration and rapid rehydration. Their erythrocytes can withstand considerable osmotic stress, responding in a manner more similar to the elliptical, nucleated erythrocytes of non-mammalian vertebrates than to the biconcave diskoidal cells typical of other mammals (Ponder, 1942; Trotter, 1956). Camel erythrocytes are highly resistant to hypotonic haemolysis (Perk, 1963; Yagil, Sod-Moriah & Meyerstein, 1974), exhibit a low rate of water transport (Naccache & Sha’afi, 1974), and are also relatively stable under hypertonic conditions, in which crenation was not observed (Yagil et al. 1974). In addition, very young camels (6 months or less) apparently possess 2 populations of erythrocytes with respect to osmotic resistance: one population with adult-type response, the other with still greater haemolytic resistance (Perk, 1966). Direct studies of the possible correlation between occurrence of MBs and osmotic resistance in camel erythrocytes would therefore appear to have potential value for understanding MB and erythrocyte function.
__________________
And also this other paper:
– Azwai SM et al. Morphological characteristics of blood cells in clinically normal adult llamas (Lama glama). Veterinarski arhiv (2007) vol. 77 (1) pp. 69-79
hrcak.srce.hr/index.php?show=clanak&id_clanak_jezik=39723
vef.unizg.hr/vetarhiv/papers/2007-77-1-9.pdf
South American camelids have evolved at elevations of 4000 meters or more and are therefore well adapted to high altitudes. The unique morphological features of llama erythrocytes play a vital role in their ability to transport oxygen to the tissue adequately at these elevations without increasing their erythrocyte mass. The leukogram is characterized by a high total leukocyte count accompanied by high neutrophil and eosinophil counts (REYNAFARJE et al., 1968).
[ . . . ]
Erythrocytes. Erythrocytes were numerous (10.3 to 15.0 ×106/μL), small (7.32 ± 0.95 × 3.9 ± 0.52 μm), flat, elliptical, lacked central pallor and their outlines were less distinct than usual (Fig. 1). Their diameters varied from 5.74 to 11.48 μm and 3.43 to 5.74 μm for the greater and lesser diameters, respectively. A few red blood cells were folded and slight anisocytosis was common. The calculated MCV was low, while MCHC was high. Reticulocytes and nucleated erythrocytes were larger and more rounded than mature ones. Faint lines in the form of a ring bordering the cell membrane or figure eight occupying the whole cell were observed in 2.7% of the llamoid erythrocytes in brilliant cresyl blue stained blood smears (Fig. 2). These lines were identified as Cabot’s rings. Polychromatic rubricytes, metarubricytes (0.8/100WBC) and cells containing Howell-Jolly bodies were observed in May-Grünwald-Giemsa stained blood smears. Distribution of these cells was 20%, 74% and 6%, respectively, among 100 nucleated erythrocytes.
Platelets. Platelet count ranged from 2.1 to 8.0 ×105/μL. They were small, with a mean diameter of 2.5 μm and a range of 1.72 to 4.59 μm. Platelets were round to irregular in shape, with pinkish cytoplasm containing azurophilic granules (Fig. 1).
[ . . .]
Discussion
The mean and range values of haematological parameters examined in this study were within the ranges established previously for clinically normal llamas (Lama glama) (AL-IZZI et al., 2004).
Llamoid erythrocytes were small (7.32 ± 0.95 × 3.9 ± 0.52 μm), elliptical, flat and their counts obtained in the present study (10.3 to 15.0 ×106/ μL) were higher than those in other domestic animals (FELDMAN et al., 2000). The flat shape and the presence of the few folded erythrocytes were attributed to the low thickness to diameter ratio of llama red blood cells (VAN HOUTEN et al., 1992). The small volume resulted in a high concentration of erythrocytes for any given PCV. The high MCHC of llama (38.6 to 48.0 g/dL) in comparison with those in other species might be due to the flat nature of the erythrocytes, which allowed more space for haemoglobin molecules to increase their efficiency for carrying oxygen at high altitude (HAWKEY, 1975). It was evident that llama bone marrow, unlike other ruminants except dromedary camels (MOORE, 2000), was normally releasing immature erythrocytes, including polychromatic rubricytes, metarubricytes (0.8%) and reticulocytes (0.4%) into circulation.
[ . . . ]
In general, the unique elliptical, flat camelid erythrocytes facilitate their movement in capillaries at times of dehydration in arid areas, (SMITH et al., 1979; SMITH et al., 1980) minimizing the likelihood of sludging.
Very neat information on red blood cell shape and function in the camelids. Thanks for finding this for us, Zephyr López Cervilla .
Rajini Rao,
So do you think that may exist some relation between the eliptic and flattened shape of red blood cells that is characteristic of camelids and the independent development of their HCAb’s? If so, what possible relation?
Now I know where I’ve seen that face before: http://goo.gl/kGyHj
from http://goo.gl/Ah3oF
This photo looks like the singer in “Flock of Seagulls”.
I thought it looked like the Mayor of London – maybe it’s a look.
Jimmy Shepard He really must share the resemblance because you are the third person in this thread to make that connection! I had not heard that song before, it was quite a revelation 😀
The flaxen blond does the trick, Kevin Clift 🙂
Oh God… you are right… I didn’t see those comments. Wow!