• WATER • AGUA • VODA • PANI • SHOUEI • How do you say it?
In recognition of World Water Day, here’s some of my favorite water trivia.
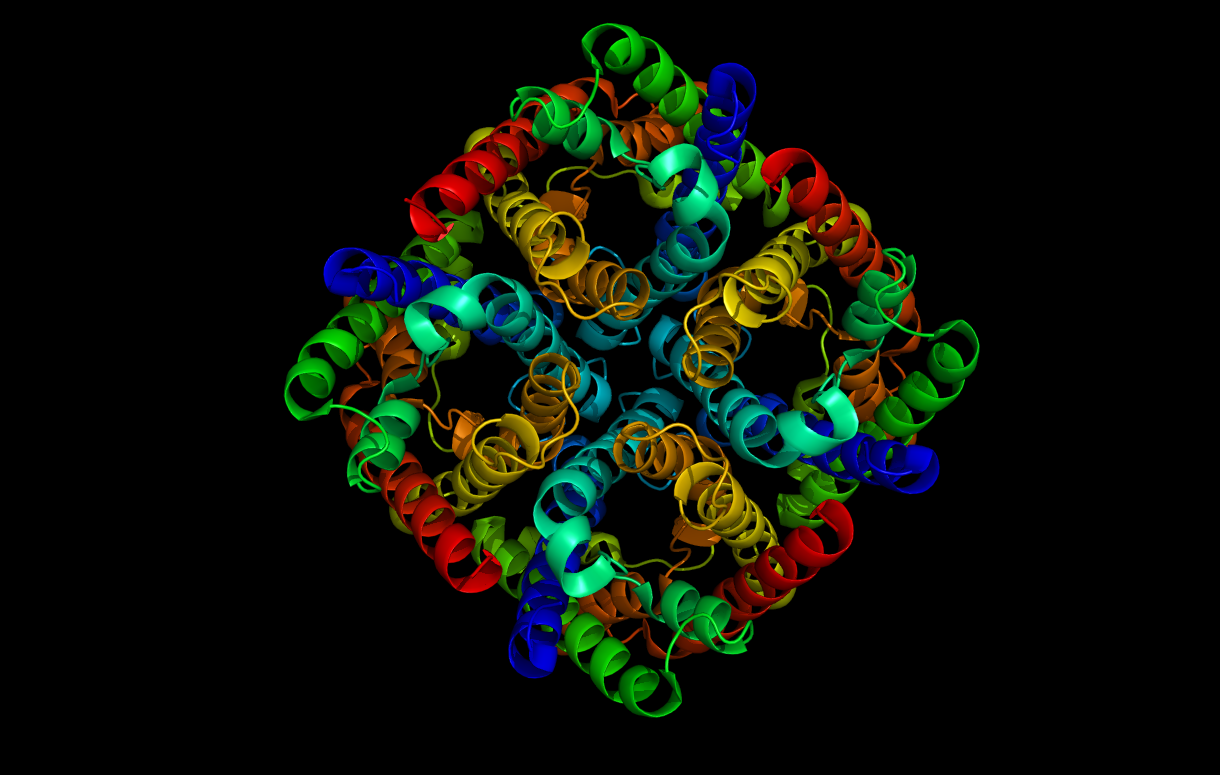
• Water Channels: If you were impressed by Google’s announcement of ultra-high-speed internet service to Kansas City at one giga-bit per second then consider this: the water channel Aquaporin moves 10,000,000,000 molecules of water each second! Aquaporin (Image 2) is a protein found in cell membranes of all plants and animals, wherever water needs to be moved.
• Water Dipoles: Although a molecule of water is neutral, it is a strong dipole with a negative (oxygen) and positive (two hydrogens) end (Image 3). This accounts for the mutual attraction between water molecules (hydrogen bonding) and explains many of its wonderful properties such as surface tension, structure of ice and high boiling point. Ion channels can be selective for a larger ion (such as K+) over a smaller one (Na+) because of the energetic costs of stripping the larger hydration shell off the smaller ion.
• The ocean is a large drop; a drop is a small ocean. Ralph Waldo Emerson (Image 4) Did you ever wonder at the thought that the same molecules of water we use today were around when the earth was formed?
• Water, water, everywhere, Nor any drop to drink. –Samuel Taylor Coleridge, The Rime of the Ancient Mariner. 97% of all water on earth is in the ocean but we can’t drink it. We need salt and we need water, so what’s the big deal? Because seawater is more concentrated than our cells, we actually lose water by osmosis and become dangerously dehydrated. Interestingly, drinking too much pure water is just as dangerous and athletes have been known to die of water toxicity. Osmosis drives balance of water across our cells (Image 1).
What are some of your favorite or weird water facts? 🙂
Many Thanks to Feisal Kamil who suggested a post relating to World Water Day!




I say it AIGUA 🙂
In what language, Víktor Bautista i Roca ?
In Catalan 🙂
Actually, I did look for the Malay translation of water but did not put it in because I thought it would confuse: Water, Air 🙂
Be proper now, Feisal Kamil 😉
Shaker Cherukuri , “Ice is forming on the tips of my wings, Unheeded warnings, I thought I thought of everything”. From Learning to Fly, PF.
In other languages I (barely) know: Swahili maji, Qazaq su.
I think in quite a few other Turkic languages is su too, as in Uyghur, Turkish, Azerbaijani or Tatar. And in many Eastern Africa Bantu languages is something similar to maji, as Kikuyu mai or Lingala mái.
Prabat parmal Aqua pura is Latin for pure water, not greek. Water in greek is νερό.
Did you ever wonder at the thought that the same molecules of water we use today were around when the earth was formed?
Is this true? I mean, as everyone in school I’ve destroyed water molecules (the typical electrolysis experiment). No new ones can be formed?
I’ve forgotten almost all Chemistry I learned at school, Feisal Kamil, but I think hydrogen in the air immediately joins oxygen to form new water molecules, but I’m not sure. I also think that the oxygen we breath is used to combine with the hydrogen byproduct of cells feeding, but I’m not sure.
nice
hi fdz
Rajini Rao Goodness gracious me. But you share interesting stuff. In South Africa we have the Comrades Maraton run over 80 km or plus 50 miles. A clever Dr, Tim Noakes wrote a book Called the Law Of Running. Here’s how this site Noakes http://goo.gl/Ze7Yq
One of the most respected researchers on hydration, Dr. Tim Noakes, studied the effects of thousands of endurance athletes and noted that the front-runners typically tend to dehydrate, while over-hydration occurs most often among middle to back-of-the-pack athletes. Both conditions lead to hyponatremia (low blood sodium), but through different processes. Excess water consumption causes what is known as “dilutional hyponatremia,” or an overly diluted level of sodium and electrolytes in the blood. This is as bad as under-hydrating in regards to increased potential for muscular cramping but has the added disadvantages of stomach discomfort, bloating, and extra urine output. And, as mentioned earlier, in some unfortunate circumstances, excess hydration can leads to severe physiological circumstances, including death.
Have fun.
MRI takes advantage of the fact that the body is approximately 75% water. It’s the protons from water that are able to be manipulated by the strong magnetic fields used in MRI. More recently, Diffusion Weighted Imaging, is an MRI technique that images the diffusion of fluid (mostly water). DWI is becoming standard clinical practice for cancer research. The neuro-guys like Diffusion Tensor Imaging, where you also image the direction of diffusion.
Nice post once again Rajini Rao
Sorry Feisal Kamil and Rajini Rao I’m trying to work so know clever link to Pink Floyd or any cool music. Maybe later.
Gnotic Pasta reminds me of when I used to work on cars with my dad. I’d make up names for parts to see if he was paying attention. Like, “I bet it will run smoother now with the new dual-poly-tootenators I installed”.
“Water is best,” wrote Greek poet Pindar in his First Olympian Ode. Pindar ranked water before gold in importance…
Interesting point about toxicity even with pure water Rajini Rao . I’m guessing we can refer to it with the TDS (ppm) level of water? There is much confusion regarding this with only one half of people saying 0-50 TDS is ideal level for drinking water.
Forgot to add my 2 cents. In Thai water is น้ำ (nom, rhymes with prom).
Edit: oops, it rhymes with prom not ham. If it rhymes with ham it means pickled sausage. ;~) This just proves that I’m not bilingual by any stretch of the imagination. Although I wish I were!
What about deuterium oxide, toxic heavy water? It’s actually nice to use when calibrating MRI because you can dilute water with D2O to lower the signal.
Forgot to add: cue Feisal Kamil for Rhyme of the Ancient Mariner or some other heavy metal but here’s some real heavy metal. http://youtu.be/JNG_Kn-m73M
Gnotic Pasta Thankfully, contrary to popular belief about drinking water, I was never over-excited about over-hydration! 😛
Thank you, Víktor Bautista i Roca , Gnotic Pasta , Chad Haney , Feisal Kamil and Sinead Woods for all those exotic words! Nothing so obvious as French or German here, of course 😉
I knew a guy trying to get people to invest in Penta-Water. What a scam.
http://www.chem1.com/CQ/pentabunk.html
It’s not like the Waterboy is saying that’s high quality H2O. BTW, The Waterboy is one of my favorite stupid movies.
http://youtu.be/VSulFZ7XJ1U
beautiful picture
Víktor Bautista i Roca and Prabat parmal , re. the amount of water in our atmosphere being constant. It was not meant to be quite that literal 🙂 As Feisal Kamil pointed out, some small amount of water may be electrolysed by lightning. Some water is brought into the atmosphere by meteorites. But the vast majority of water that is cycled (through biology) is as follows: first, plants ‘fix’ CO2 and H2O to make glucose and release oxygen by capturing energy from sunlight. Then, nearly all organisms (plants, animals, etc.) release that energy to do work, by breaking the glucose and releasing CO2 and H2O. So the water that is consumed in photosynthesis is regenerated during respiration.
ALSO, there are tons of condensation reactions in biology where two molecules come together to form a polymer. Water is typically released during that condensation. Example, when amino acids are concatenated to form a protein..it is a dehydration reaction, so water is released at each step.
Vietnamese is nước, (nuoc,pronounced like new-uk, like new book)
Kudos to +javi lamas gomez for correcting me on my inversion of electronegative (oxygen) and electropositive (hydrogen) in the dipole. I’ve edited the post!
Are there any other molecules which expand when they cool?
Matt Kuenzel , good point..this is a famous peculiarity of water. I believe some metal alloys expand upon cooling. I found this: Cubic zirconium tungstate (ZrW2O8) is a widely studied material that expands upon cooling over a wide temperature range. There are other metal tungstates and vanadates that have been studied as well. In addition, there is a naturally occuring mineral, beta-eucryptite, that expands slightly upon cooling.
Rajini Rao That is a strange property. Apparently there are also a variety of ways that water can crystallize (depending on pressure?) … the novel Ice 9 by Vonnegut uses that property.
Nice photos 🙂
Feisal Kamil implies no trans-fat! :~)
Surely some enterprising scammer has come up with zero calorie water by now? If not, we thought of it first here!
Feisal Kamil another lemming?
OMG, listen to this scam: “Usually the flavored diet waters taste like water in a dirty glass, with just a hint of fruit or flavor.” But not this great American product! “You get the satisfaction of drinking water, without the boring water taste.” Masterful marketing!
Kind of like pink lemonade, only 10x more gross and fattening. It would still be a water and beef product; no need for special labeling.
Prabat parmal , here is some information: http://www.ehow.com/about_6697335_ways-combine-hydrogen-oxygen-water.html
A beautiful video..well worth the watch for the artwork, thanks Feisal Kamil !
LOL, Mahesh Sreekandath ! The real question is how did you find this? I keep getting random likes of ancient posts and I’ve always wondered. Do people use Google search bar for topics or??
Mystery of the history explained, thanks Mahesh Sreekandath ! I’ll check out the song 🙂
G+ ate my comment again! Just in case, enjoy Faraday Feisal Kamil
Rime of the ancient mariner is an all time fav from Iron Maiden. Goodnight Feisal Kamil enjoy the weekend.